Publications
Independent at LSU
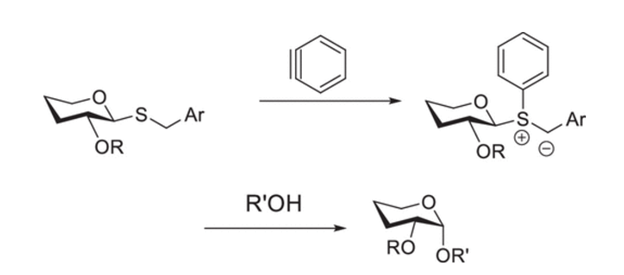
Abstract: Here, we show that the reaction of benzylchalcogenoglycosides with benzyne in the presence of alcohols results in highly 1,2-cis-selective O-glycosylation in a solvent-dependent manner. Thioglycosides, selenoglycosides, and alcohols with a range of nucleophilicities lead to a productive reaction, and unusual protecting groups, auxiliary groups, and additives are avoided.
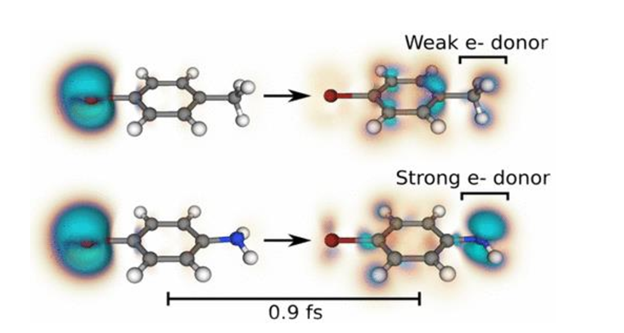
Abstract: Charge migration (CM) is a coherent attosecond process that involves the movement of localized holes across a molecule. To determine the relationship between a molecule’s structure and the CM dynamics it exhibits, we perform systematic studies of para-functionalized bromobenzene molecules (X-C6H4-R) using real-time time-dependent density functional theory. We initiate valence-electron dynamics by emulating rapid strong-field ionization leading to a localized hole in the bromine atom. The resulting CM, which takes on the order of 1 fs, occurs via an X localized → C6H4 delocalized → R localized mechanism. Interestingly, the hole contrast on the acceptor functional group increases with increasing electron-donating strength. This trend is well-described by the Hammett s value of the group, which is a commonly used metric for quantifying the effect of functionalization on the chemical reactivity of benzene derivatives. These results suggest that simple attochemistry principles and a density-based picture can be used to predict and understand CM.
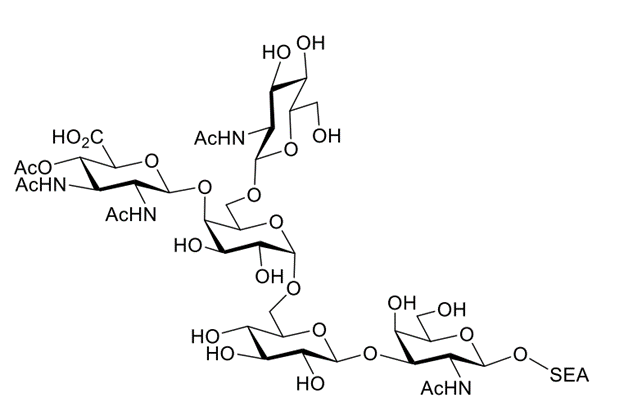
Abstract: Acinetobacter baumannii is a Gram-negative bacteria associated with drug resistance and infection in healthcare settings. An understanding of both the biological roles and antigenicity of surface molecules of this organism may provide an important step in the prevention and treatment of infection through vaccination or the development of monoclonal antibodies. With this in mind, we have performed the multistep synthesis of a conjugation-ready pentasaccharide O-glycan from A. baumannii with a longest linear synthetic sequence of 19 steps. This target is particularly relevant due to its role in both fitness and virulence across an apparently broad range of clinically relevant strains. Synthetic challenges include formulating an effective protecting group scheme as well as the installation of a particularly difficult glycosidic linkage between the anomeric position of a 2,3-diacetamido-2,3-dideoxy-D-glucuronic acid and the 4-position of D-galactose.
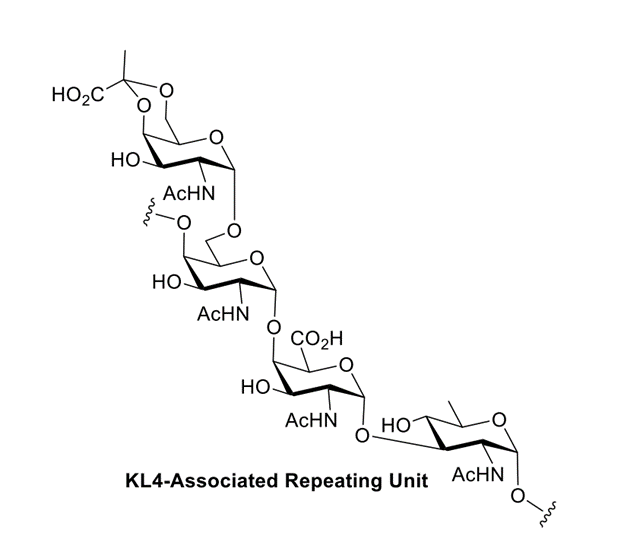
Abstract: Chemical synthetic efforts have resulted in the preparation of the assigned tetrasaccharide repeating subunit from the Acinetobacter baumannii KL4-associated capsular polysaccharide. A convergent synthetic strategy hinging on a 1,2-cis-selective [2+2] glycosylation to generate the fully protected tetrasaccharide was key to the success of this synthesis.
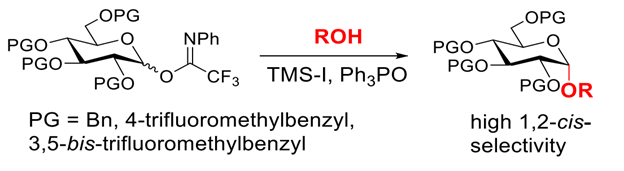
Abstract: Here, we demonstrate that substitution of the benzyl groups of glucosyl imidate donors with trifluoromethyl results in a substantial increase in 1,2-cis-selectivity when activated with TMS-I in the presence of triphenylphosphine oxide. Stereoselectivity is dependent on the number of trifluoromethyl groups (4-trifluoromethylbenzyl vs. 3,5-bis-trifluoromethylbenzyl). Particularly encouraging is that we observe high 1,2-cis-selectivity with reactive alcohol acceptors.
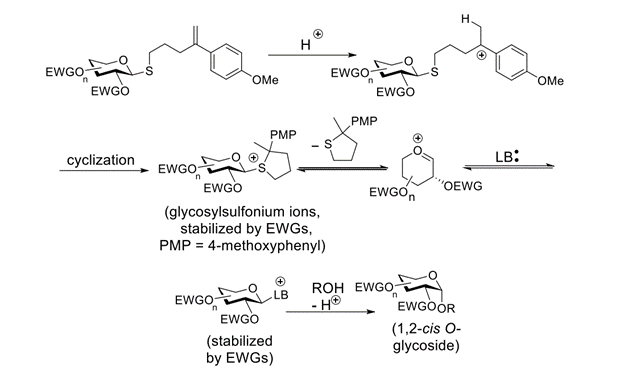
Abstract: We report on our initial results from a systematic effort to implement electron-withdrawing protecting groups and Lewis-basic solvents/additives as an approach to 1,2-cis(a)-selective O-glucosylation. 1,2-Cis-selective O-glucosylations are reported with thioglucosides and glucosyl trichloroacetimidates and a range of acceptors. A correlation between electron-withdrawing effects and 1,2-cis selectivity has been established. This phenomenon may prove to be broadly applicable in the area of chemical O-glycosylation.
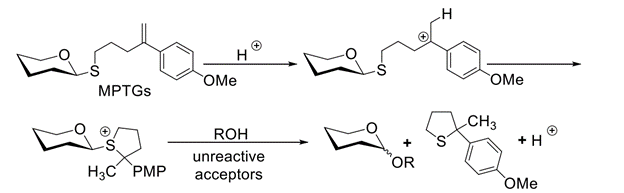
Abstract: MPTGs (4-(p-methoxyphenyl)-4-pentenylthioglycosides) undergo acid-catalyzed O-glycosylation with a range of alcohol acceptors in the presence of 10 mol % triflic acid at room temperature. Particularly encouraging is the reactivity of MPTGs toward unreactive acceptors. MPTGs can be synthesized from the requisite vinyl bromides using the Suzuki reaction, and this chemistry can be leveraged toward a “latent-active” strategy for oligosaccharide synthesis.
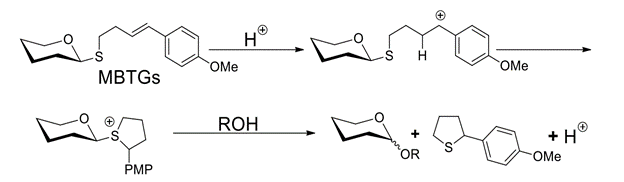
Abstract: Two classes of thioglycoside, 4-(4-methoxyphenyl)-3-butenylthioglycosides (MBTGs) and 4-(4-methoxyphenyl)-4-pentenylthioglycosides (MPTGs), undergo acid-catalyzed O-glycosylations with a range of sugar- and non-sugar alcohols at 25oC. Electron density at styrene alkene is critical for reactivity while sugar protecting group patterns have a minimal effect. To contrast with most methods for thioglycoside activation, acid-catalyzed activation of MBTGs is compatible with electroneutral alkenes.
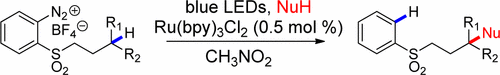
Abstract: Visible-light irradiation ofortho-diazoniaphenyl alkyl sulfones in the presence of Ru(bpy)32+results in remote Csp3–H functionalization. Key mechanistic steps in these processes involve intramolecular hydrogen atom transfer from Csp3–H bonds to aryl radicals to generate alkyl/benzyl radicals. Subsequent polar crossover occurs by single-electron oxidation of the alkyl/benzyl radicals to carbenium ions that then intercept nucleophiles. We have developed remote hydroxylations, etherifications, an amidation, and C–C bond formation processes using this strategy.
10. Quarels, R.D.; Zhai, X.; Kuruppu, N.; Hedlund, J.K.; Ellsworth, A.A.; Walker, A.V.; Garno, J.C.; Ragains, J.R. Application of Visible-Light Photosensitization to Form Alkyl-Radical-Derived Thin Films on Gold. Beilstein J. Nanotechnol. 2017, 8, 1863-1877. DOI: 10.3762/bjnano.8.187.
![equations of Visible-light irradiation of phthalimide esters in the presence of the photosensitizer [Ru(bpy)3]2+ and the stoichiometric reducing agent benzyl nicotinamide results in the formation of alkyl radicals under mild conditions](/ragains/images/nanotechnol.jpg)
Abstract: Visible-light irradiation of phthalimide esters in the presence of the photosensitizer [Ru(bpy)3]2+and the stoichiometric reducing agent benzyl nicotinamide results in the formation of alkyl radicals under mild conditions. This approach to radical generation has proven useful for the synthesis of small organic molecules. Herein, we demonstrate for the first time the visible-light photosensitized deposition of robust alkyl thin films on Au surfaces using phthalimide esters as the alkyl radical precursors. In particular, we combine visible-light photosensitization with particle lithography to produce nanostructured thin films, the thickness of which can be measured easily using AFM cursor profiles. Analysis with AFM demonstrated that the films are robust and resistant to mechanical force while contact angle goniometry suggests a multilayered and disordered film structure. Analysis with IRRAS, XPS, and TOF SIMS provides further insights.
9. Spell, M.L.; Deveaux, K.; Bresnahan, C.G.; Ragains, J.R. "O-Glycosylation Enabled by Remote Activation." Synlett. 2017, 28 (7), 751-761. DOI: 10.1055/s-0036-158894.
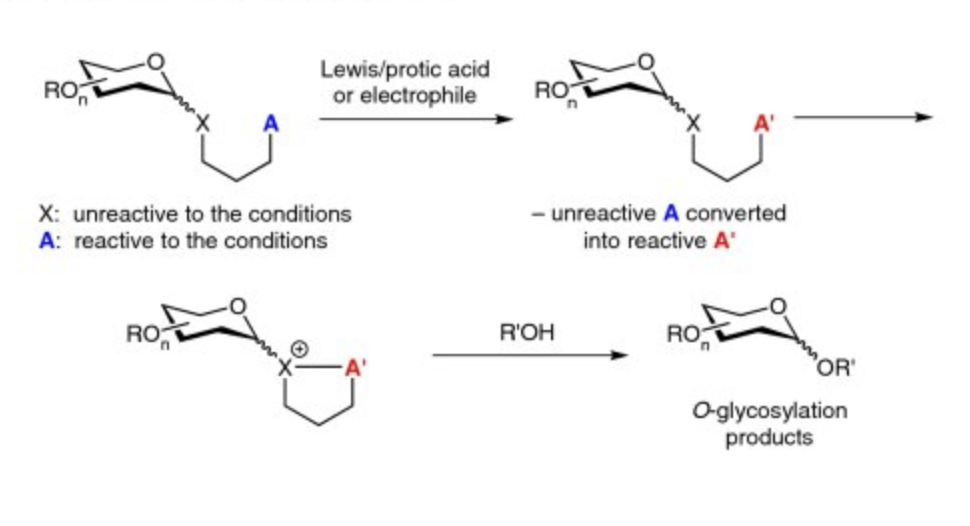
Abstract: O-Glycosylation is a critically important and recurring step in the synthesis of oligosaccharides and other natural and non-natural products. While many approaches to O-glycosylation have been reported, those strategies involving remote activation are distinguished by the mildness and orthogonality that they often engender. As a result, O-glycosylation using remote activation strategies has been utilized successfully in the synthesis of complex molecules that include oligosaccharides and macrolides. Herein, we discuss a number of contributions that have been made to this area since the 1970s. This includes our own recent contribution involving the visible-light activation of 4-para-methoxyphenyl-3-butenylthioglycosides towardO-glycosylation in the presence of Umemoto's reagent.
8. Spell, M.L.; Deveaux, K.; Bresnahan, C.G.; Bernard, B.L.; Sheffield, W.; Kumar, R.; Ragains, J.R. "A Visible Light-Promoted O-Glycosylation with a Thioglycoside Donor." Angew. Chem. Int. Ed. 2016, 55, 6515-6519.
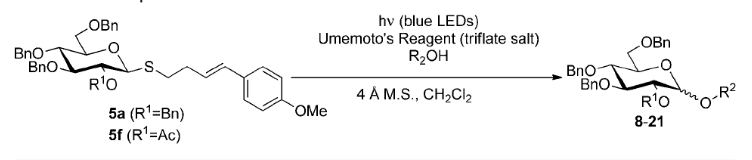
Abstract: Visible-light irradiation of 4-p-methoxyphenyl-3-butenylthioglucoside donors in the presence of Umemoto's reagent serves as an exceptionally mild and orthogonal approach to O-glycosylation. Alkyl- and arylthioglycosides not containing the p-methoxystyrene moiety are inert under these conditions, and evidence suggests that an intervening electron donor–acceptor complex is necessary for reactivity.
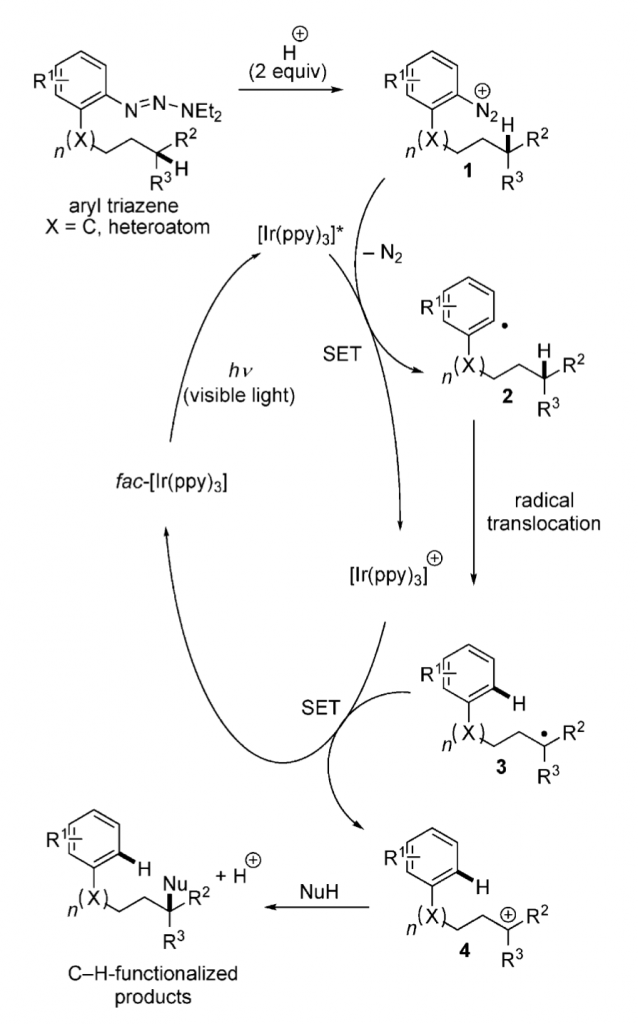
Abstract: Mild conditions have been developed for the remote functionalization of aliphatic CH bonds through radical translocation and oxidation of the resulting radical to the carbocation as a prerequisite to nucleophilic attack. The employment of fac-[Ir(ppy)3] together with the Tzo group facilitates the site-selective replacement of inert CH bonds with a COH group.
Abstract: Visible light photoredox catalysis was combined with immersion particle lithography to prepare polynitrophenylene organic films on Au(111) surfaces, forming a periodic arrangement of nanopores. Surfaces masked with mesospheres were immersed in solutions of p-nitrobenzenediazonium tetrafluoroborate and irradiated with blue LEDs in the presence of the photoredox catalyst Ru(bpy)3(PF6)2 to produce p-nitrophenyl radicals that graft onto gold substrates. Surface masks of silica mesospheres were used to protect small, discrete regions of the Au(111) surface from grafting. Nanopores were formed where the silica mesospheres touched the surface; the mask effectively protected nanoscopic local areas from the photocatalysis grafting reaction. Further reaction of the grafted arenes with aryl radicals resulted in polymerization to form polynitrophenylene structures with thicknesses that were dependent on both the initial concentration of diazonium salt and the duration of irradiation. Photoredox catalysis with visible light provides mild, user-friendly conditions for the reproducible generation of multilayers with thicknesses ranging from 2 to 100 nm. Images acquired with atomic force microscopy (AFM) disclose the film morphology and periodicity of the polymer nanostructures. The exposed sites of the nanopores provide a baseline to enable local measurements of film thickness with AFM. The resulting films of polynitrophenylene punctuated with nanopores provide a robust foundation for further chemical steps. Spatially selective binding of mercaptoundecanoic acid to exposed sites of Au(111) was demonstrated, producing a periodic arrangement of thiol-based nanopatterns within a matrix of polynitrophenylene.
5. McKnight, K.; Hoang, H.D.; Prasain, J.K.; Brown, N.; Vibbert, J.; Hollister, K.A.; Moore, R.; Ragains, J.R.; Reese, J.; Miller, M.A. "Neurosensory Perception of Environmental Cues Modulates Sperm Motility Critical for Fertilization." Science 2014, 344, 754-757.
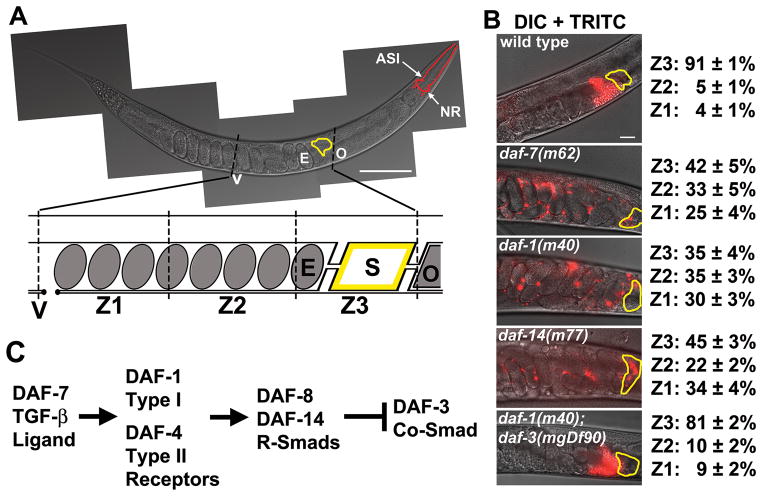
Environmental exposures affect gamete function and fertility, but the mechanisms are poorly understood. Here, we show that pheromones sensed by ciliated neurons in the Caenorhabditis elegans nose alter the lipid microenvironment within the oviduct, thereby affecting sperm motility. In favorable environments, pheromone-responsive sensory neurons secrete a transforming growth factor–β ligand called DAF-7, which acts as a neuroendocrine factor that stimulates prostaglandin-endoperoxide synthase [cyclooxygenase (Cox)]–independent prostaglandin synthesis in the ovary. Oocytes secrete F-class prostaglandins that guide sperm toward them. These prostaglandins are also synthesized in Cox knockout mice, raising the possibility that similar mechanisms exist in other animals. Our data indicate that environmental cues perceived by the female nervous system affect sperm function.
Abstract: A visible-light-promoted method for the selenofunctionalization (and tellurofunctionalization) of alkenes has been developed. This method obviates the prepreparation of moisture-sensitive chalcogen electrophiles. The experimental setup is simple, and superior yields are obtained in the case of selenofunctionalization (up to 99%) while moderate to good yields are obtained in the case of tellurofunctionalization (53–75%). A variety of intra- and intermolecular processes and a short synthesis of the Amaryllidaceae alkaloid (±)-γ-lycorane are demonstrated with this method.
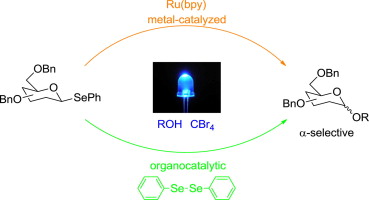
Abstract: Exceptionally mild procedures for the visible light photocatalytic activation of selenoglycoside donors in the presence of alcohol acceptors have been developed. This process is demonstrated with both 1-phenylselenyl-2,3,4,6-tetra-O-benzyl glucoside and 1-phenylselenyl-2,3,4,6-tetra-O-benzyl galactoside. Catalysis is effected with both metal (Ru(bpy)3) and organocatalysts (diphenyldiselenide). Reactions afford, in all cases, primarily the α-anomers with selectivities that vary with solvent. This represents the first example of a visible light-promoted O-glycosylation.
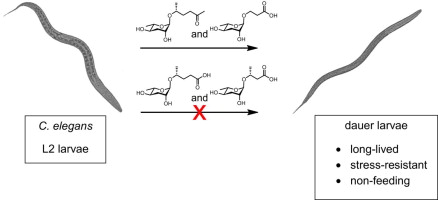
Abstract: The nematode Caenorhabditis elegans secretes ascarosides, structurally diverse derivatives of the 3,6-dideoxysugar ascarylose, and uses them in chemical communication. At high population densities, specific ascarosides, which are together known as the dauer pheromone, trigger entry into the stress-resistant dauer larval stage. In order to study the structure–activity relationships for the ascarosides, we synthesized a panel of ascarosides and tested them for dauer-inducing activity. This panel includes a number of natural ascarosides that were detected in crude pheromone extract, but as yet have no assigned function, as well as many unnatural ascaroside derivatives. Most of these ascarosides, some of which have significant structural similarity to the natural dauer pheromone components, have very little dauer-inducing activity. Our results provide a primer to ascaroside structure–activity relationships and suggest that slight modifications to ascaroside structure dramatically influence binding to the relevant G protein-coupled receptors that control dauer formation.
Abstract: Entomopathogenic nematodes survive in the soil as stress-resistant infective juveniles that seek out and infect insect hosts. Upon sensing internal host cues, the infective juveniles regurgitate bacterial pathogens from their gut that ultimately kill the host. Inside the host, the nematode develops into a reproductive adult and multiplies until unknown cues trigger the accumulation of infective juveniles. Here, we show that the entomopathogenic nematode Heterorhabditis bacteriophora uses a small-molecule pheromone to control infective juvenile development. The pheromone is structurally related to the dauer pheromone ascarosides that the free-living nematode Caenorhabditis elegans uses to control its development. However, none of the C. elegans ascarosides are effective in H. bacteriophora, suggesting that there is a high degree of species specificity. Our report is the first to show that ascarosides are important regulators of development in a parasitic nematode species. An understanding of chemical signaling in parasitic nematodes may enable the development of chemical tools to control these species.